Abstract
Background: Co-deletion of 1p and 19q marks a diffuse glioma subtype associated with relatively favorable overall survival; however, heterogeneous clinical outcomes are observed within this category.
Methods: We assembled gene expression profiles and sample annotation of 374 glioma patients carrying the 1p/19q co-deletion. We predicted 1p/19q status using gene expression when annotation was missing. A first cohort was randomly split into training (n = 170) and a validation dataset (n = 163). A second validation set consisted of 41 expression profiles. An elastic-net penalized Cox proportional hazards model was applied to build a classifier model through cross-validation within the training dataset.
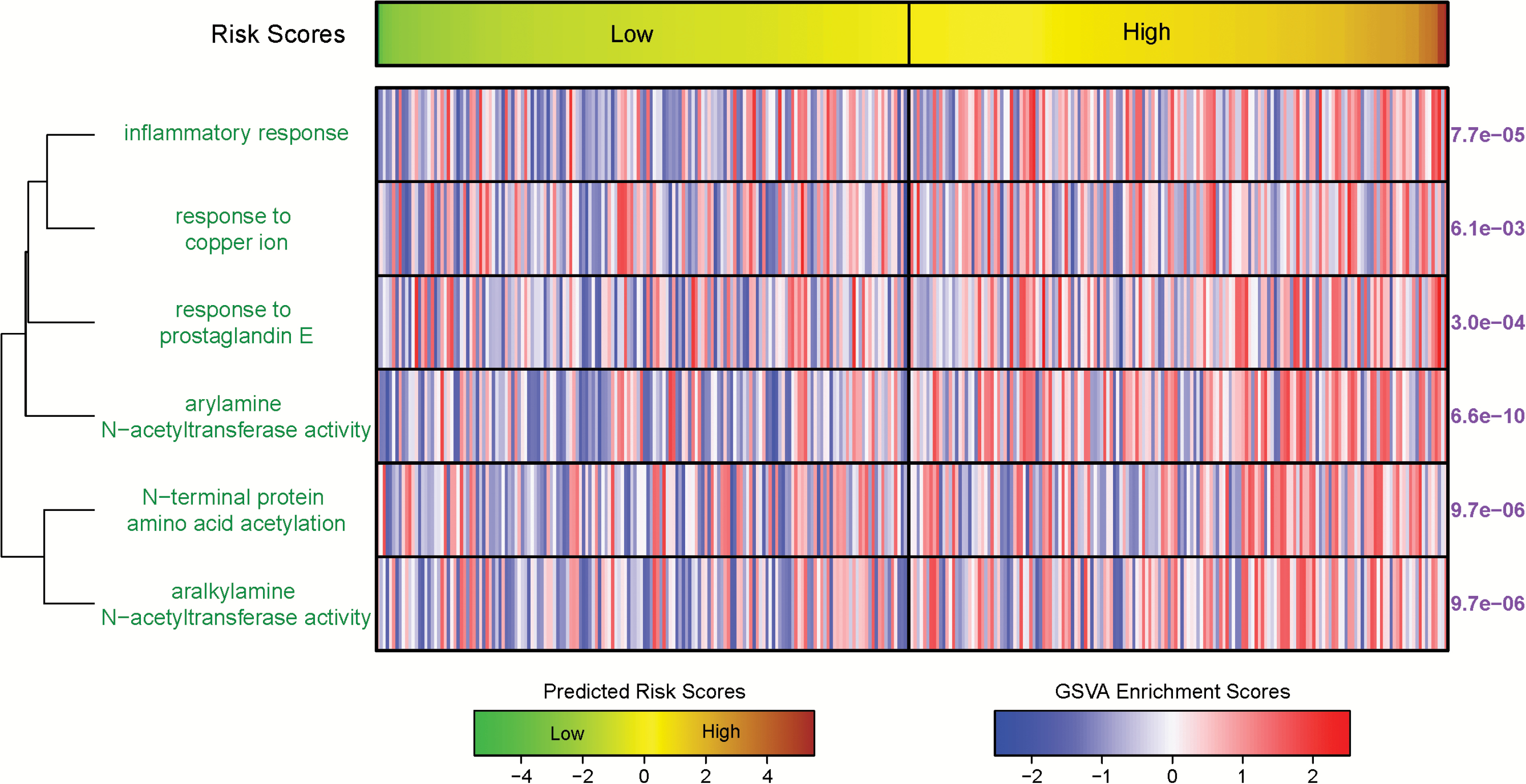
Results: The selected 35-gene signature was used to identify high-risk and low-risk groups in the validation set, which showed significantly different overall survival (P = .00058, log-rank test). For time-to-death events, the high-risk group predicted by the gene signature yielded a hazard ratio of 1.78 (95% confidence interval, 1.02-3.11). The signature was also significantly associated with clinical outcome in the The Cancer Genome Atlas (CGA) IDH-mutant 1p/19q wild-type and IDH-wild-type glioma cohorts. Pathway analysis suggested that high risk was associated with increased acetylation activity and inflammatory response. Tumor purity was found to be significantly decreased in high-risk IDH-mutant with 1p/19q co-deletion gliomas and IDH-wild-type glioblastomas but not in IDH-wild-type lower grade or IDH-mutant, non-co-deleted gliomas.
Conclusion: We identified a 35-gene signature that identifies high-risk and low-risk categories of 1p/19q positive glioma patients. We have demonstrated heterogeneity amongst a relatively new glioma subtype and provided a stepping stone towards risk stratification.